HempMeds RSHO CBD
 Real Scientific Hemp Oil (RSHO®) CBD
Real Scientific Hemp Oil (RSHO®) CBD
Full-Spectrum Organic Hemp-Derived Cannabidiol Capsules
(For available discounts off the MedHemps List Price please use our Contact page and send us an email inquiry)
Hemp is a cannabis plant that contains multiple beneficial compounds called cannabinoids. The major cannabinoids are CBD (cannabidiol) and THC (tetrahydrocannabinol).
Hemp has high concentrations of CBD (non-psychoactive) and only trace amounts of THC (psychoactive), therefore CBD has only medicinal and no recreational utility.
In 2003 the U.S. Government, Department of Health and Human Services, obtained U.S. Patent #6630507 titled Cannabinoids as Antioxidants and Neuroprotectants. The patent describes cannabinoids as "useful in the treatment and prophylaxis of wide variety of oxidation associated diseases, such as ischemic, age-related, inflammatory and autoimmune diseases."
CBD is just one of over 85 cannabinoids discovered so far in the cannabis plant and is widely regarded as the cannabinoid with the most medicinal potential by researchers.
Extensive research indicates CBD has the following promising therapeutic actions: anti-inflammatory, anti-convulsive, anti-nausea, anti-anxiety, anti-oxidation, anti-tumoral. In fact, studies have shown that CBD is more protective against neurotoxicity than either ascorbate (Vitamin C) or α-tocopherol (Vitamin E), indicating it to be a potent antioxidant.
RSHO (Real Scientific Hemp Oil) contains CBD that is considered a dietary supplement, is legal to purchase in all 50 U.S. states, and contains less than 0.3% THC by weight which exempts it from the Federal Controlled Substances Act prohibition on marijuana. Also, the U.S. 2018 Farm Bill officially removed all Hemp-derived extracts including CBD from the federal list of controlled substances.
RSHO contains concentrated CBD and other cannabinoids, Omega 3 & 6 essential fatty acids, and multiple vitamins and minerals such as iron, calcium, magnesium, vitamins A, B, C, and E.
RSHO is sourced from a Certified Organic family-owned farm in the Netherlands that has been awarded an Organic Certification from Skal Biocontrole, the control authority in Holland recognized by the European Union (EU). The hemp cultivated on this farm is grown under highest standards in accordance with European organic regulations and the Dutch Agricultural Quality Act.
RSHO is Triple Lab Tested® for contaminants such as herbicides, pesticides, chemical fertilizers, mold, fungus, bacteria, or heavy metals, and to ensure purity, consistency, and accurate potency of cannabinoid content.
RSHO is the first legally available CBD product in major foreign markets like Argentina, Brazil, Mexico, and Paraguay because of its validated quality, purity and potency.
RSHO's manufacturer, HempMeds, has a Certification Seal from the U.S. Hemp Authority, meaning the product has met several stringent industry standards laid out for quality and safety. The certification is intended to educate hemp farmers and producers about Food and Drug Administration Current Good Manufacturing Practices (cGMP) and Good Agricultural Practices (GAP) to guarantee that consistent quality is achieved.
RSHO and other company CBD products have been in clinical studies or have been prescribed for the treatment of pediatric epilepsy, restless leg syndrome, amyotrophic lateral sclerosis, psoriasis, drug-induced adult psychosis, opioid addiction, glaucoma, dry eye, irritable bowel syndrome, early-stage Alzheimer’s disease, and the spasticity of multiple sclerosis.
RSHO Gold CBD products are included in the prestigious drug and supplement database - the PDR (Physician's Desk Reference now referred to as Prescriber's Digital Reference) used by doctors. pharmacists, and other health care practitioners: "RSHO-Goldâ„¢ may be useful as an adjunctive supplement to support healthy immune response and homeostasis, promote and maintain healthy mood, energy, sleep, pain response possibly via the modulation of endocannabinoid system (ESC), which is known as a vital factor in the regulation of homeostasis, symptomatic relief and overall metabolic functions."
Most workplace and sports drug screens target tetrahydrocannabinol (THC). RSHO-Xâ„¢ (containing zero THC) is the first cannabis-based product to qualify as safe for World Anti-Doping standards and can safely be used by Olympic athletes. RSHO-Xâ„¢ has also undergone strict FDA Q1A(R2) stability testing to ensure that the integrity of RSHO-Xâ„¢ will not be compromised while in transportation or while being stored for future use.
RSHO Green Label is unfiltered full-spectrum hemp oil naturally high in raw cannabinoids. The capsules have 25mg of CBD and 200mg of turmeric/white willow bark (anti-inflammatory).
DOSAGE: Every person has a unique metabolism and body physiology, but general CBD (cannabidiol) guidelines suggest a health maintenance or mild condition specific treatment daily dosage is up to 25mg, mild-moderate condition specific treatment daily dosage ranges from 25-50mg, moderate to severe condition specific treatment daily dosage ranges from 50-100mg, and severe condition specific treatment daily dosage exceeds 100mg. Usually it is best to split higher daily dosages into 2-3 equally spaced applications (such as three times daily is once every eight hours). All persons should start with a smaller daily dosage and increase gradually only as proven tolerated (no undue side-effects). The National Institutes of Health, U.S. National Library of Medicine states that CBD (cannabidiol) doses of up to 300 mg daily have been taken by mouth safely for up to 6 months.
NOTE: Women who are pregnant and/or breast feeding should avoid taking any supplement or substance without a specific recommendation by their physician.
SIDE EFFECTS: Recent studies have shown that CBD (cannabidiol) has a favorable safety profile. The most commonly reported side effects were tiredness, diarrhea, and changes of appetite/weight. In comparison with pharmaceutical drugs used for the treatment of similar medical conditions for which people often supplement CBD, a better side effect profile can be expected by CBD. At very high doses of 10mg CBD per kg of body weight per day (equivalent to 453mg CBD/day for a 100 lbs. adult or 680mg CBD/day for a 150 lbs. adult) a very small percentage of people (1.3%) experienced transaminase (liver enzyme) elevation.
DRUG INTERACTIONS: CBD (cannabidiol) may decrease how quickly the liver breaks down some medications. Using CBD with some medications that are broken down by the liver may increase the effects and/or side effects of some medications. If you take any medications that are changed by the liver, please consult with your prescribing physician before taking CBD - particularly at higher dosages. CBD may have drug interactions with Diazepam (Valium) which is used to treat anxiety, muscle spams, and seizures, and with Clobazam (Onfi) which is used to treat seizures caused by Lennox-Gastaut syndrome.
CBD (Cannabidiol) Education
a. What You Should Know About CBD
b. How CBD Works
c. Treating Neuropathic Pain (neuritis, neuralgia, radiculopathy)
d. SARS-CoV-2
a. What You Should Know About CBD (cannabidiol) 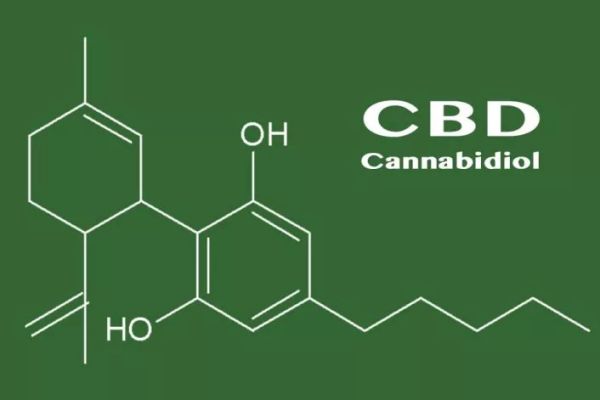
The genus Cannabis plant is an annual, dioecious, flowering herb. Indica and Sativa are the two common Cannabis species. Cannabis plants produce a large number of chemical compounds called cannabinoids, of which THC (tetrahydrocannabinol) and CBD (cannabidiol) are two of the most predominant.
THC is the psychotropic, euphoric, intoxicating component of cannabis, while CBD is a non-psychotropic, non-euphoric, and non-intoxicating cannabinoid. THC directly interacts with CB1 receptors in the brain and nerve system and CBD indirectly modulates CB1 receptors and CB2 receptors in the immune and lymphoid system and organs.
A variety of the Cannabis Sativa plant species that is bred for high concentrations of CBD and trace concentrations of THC is called Hemp, or Industrial Hemp. Page 698 of the U.S. Farm Bill (Agricultural Act of 2014), in SEC. 7606. LEGITIMACY OF INDUSTRIAL HEMP RESEARCH, under (b) Definitions states (2) INDUSTRIAL HEMP—The term ‘‘industrial hemp’’ means the plant Cannabis sativa L. and any part of such plant, whether growing or not, with a delta-9 tetrahydrocannabinol (THC) concentration of not more than 0.3 percent on a dry weight basis. Section 763 of The Omnibus Appropriations Act of 2016 goes on to state: “None of the funds made available by this act or any other act may be used… to prohibit the transportation, processing, sale or use of industrial hemp that is grown or cultivated in accordance with section 7606 of the Agricultural Act of 2014, within or outside the State in which the industrial hemp is grown or cultivated.â€
In 2003 the US Government, Department of Health and Human Services (HHS) , obtained U.S. Patent #6630507, “Cannabinoids as Antioxidants and Neuroprotectants.†Researchers at the National Institutes of Health discovered that non-psychotropic compounds in cannabis may have antioxidant properties that could be beneficial in the treatment of certain neurological diseases. The patent states: "Cannabinoids have been found to have antioxidant properties, unrelated to NMDA receptor antagonism. This new found property makes cannabinoids useful in the treatment and prophylaxis of wide variety of oxidation associated diseases, such as ischemic, age-related, inflammatory and autoimmune diseases. The cannabinoids are found to have particular application as neuroprotectants, for example in limiting neurological damage following ischemic insults, such as stroke and trauma, or in the treatment of neurodegenerative diseases, such as Alzheimer’s disease, Parkinson’s disease and HIV dementia. Non-psychoactive cannabinoids, such as cannabidiol, are particularly advantageous to use because they avoid toxicity that is encountered with psychoactive cannabinoids at high doses useful in the method of the present invention. A particular disclosed class of cananbinoids useful as neuroprotective antioxidants is formula (1) wherein the R group is independently selected from the group consisting of H, CH3 and COCH3."
The federal government's official position on Cannabis has historically ranged from outdated to overreaching. As mentioned above, the U.S. Department of Health and Human Services (HHS) holds a patent on cannabinboid compounds as antioxidants and neuroprotectants. The U.S. Food and Drug Administration (FDA) maintains that specific components of cannabis (such as the major cannabinoids THC and CBD) have medical value, but the cannabis plant itself does not. Not surprisingly, this position favors the biotech and pharmaceutical model of isolating and patenting single molecular compounds which is considered a more controlled method of conducting research and prescribing medication and supplements. It should be noted, however, that single molecule medicine may not always be the best way to benefit from cannabis therapeutics. Like most plants and herbs, Cannabis contains multiple compounds (flavonoids, terpenes, and minor cannabinoids) that when combined could create a more holistic synergistic effect (CBD isolate vs. full-spectrum Hemp Oil).
On August 9, 2017, the FDA sent out a request to interested persons to submit comments concerning abuse potential, actual abuse, medical usefulness, trafficking, and impact of scheduling changes on availability for medical use of 17 "drug substances" including cannabidiol (CBD), for consideration in preparing a response from the United States to the World Health Organization (WHO) regarding the abuse liability and diversion of these drugs. The FDA stated that one of the active cannabinoids identified in cannabis, CBD, "has been shown to be beneficial in experimental models of several neurological disorders, including those of seizure and epilepsy."
On December 14, 2016 the U.S. Drug Enforcement Agency (DEA) established a new Drug Code (7350) for marijuana extract including CBD “to track quantities of this material separately from quantities of marijuana," in part because "The United Nations Conventions on international drug control treats extracts from the cannabis plant somewhat differently than marijuana or tetrahydrocannabinols." However, the DEA does not treat "extracts" such as CBD any differently than whole plant marijuana and THC, meaning the DEA considers CBD a Schedule I controlled substance. Although a DEA spokesperson has stated that the new drug coding of "marijuana extracts", including CBD, is an internal accounting mechanism to get more accurate information about research being conducted with CBD in particular, it is important to understand the implications of scheduling a substance. Under the Controlled Substances Act, a Schedule I drug has a high abuse potential, no medical use, severe safety concerns, and includes narcotics such as Heroin, LSD, and cocaine. Given that cannabidiol (CBD) is non-psychoactive, non-addictive, has no known toxicity level, has demonstrated medical use across a wide range of conditions, is patented by the HHS as an anti-oxidant and neuroprotectant, and is cited by the FDA to be beneficial in experimental models of several neurological disorders, including those of seizure and epilepsy, it is unimaginable that the DEA ever considered CBD a scheduled controlled substance.
The World Health Organization (WHO), Expert Committee on Drug Dependence (ECDD), has stated:
"There is increased interest from Member States in the use of cannabis for medical indications including for palliative care. Responding to that interest and increase in use, WHO has in recent years gathered more robust scientific evidence on therapeutic use and side effects of cannabis and cannabis components. To that end, the ECDD did an initial review of a cannabis compound called cannabidiol (CBD). Recent evidence from animal and human studies shows that its use could have some therapeutic value for seizures due to epilepsy and related conditions. Current evidence also shows that cannabidiol is not likely to be abused or create dependence as for other cannabinoids (such as Tetra Hydro Cannabinol (THC), for instance). The ECDD therefore concluded that current information does not justify scheduling of cannabidiol and postponed a fuller review of cannabidiol preparations to May 2018, when the committee will undertake a comprehensive review of cannabis and cannabis related substances."
At it's Expert Committee on Drug Dependence Fortieth Meeting, Geneva, 4-7 June 2018, The World Health Organization issued the following summary report: "Cannabidiol (CBD) is one of the naturally occurring cannabinoids found in cannabis plants. It is a 21-carbon terpenophenolic compound which is formed following decarboxylation from a cannabidiolic acid precursor, although it can also be produced synthetically. In experimental models of abuse liability, CBD appears to have little effect on conditioned place preference or intracranial self-stimulation. In an animal drug discrimination model CBD failed to substitute for THC. In humans, CBD exhibits no effects indicative of any abuse or dependence potential. CBD has been demonstrated as an effective treatment of epilepsy in several clinical trials, with one pure CBD product (Epidiolex®) with completed Phase III trials and under current review for approval in the U.S. There is also preliminary evidence that CBD may be a useful treatment for a number of other medical conditions. There is unsanctioned medical use of CBD based products with oils, supplements, gums, and high concentration extracts available online for the treatment of many ailments. CBD is generally well tolerated with a good safety profile. Reported adverse effects may be as a result of drug-drug interactions between CBD and patients’ existing medications. Several countries have modified their national controls to accommodate CBD as a medicinal product. To date, there is no evidence of recreational use of CBD or any public health-related problems associated with the use of pure CBD."
Consider that there are 33 U.S. states, and the District of Columbia, that have legalized medical marijuana, and the remaining 17 U.S. states all have laws allowing limited use of CBD. That's 50 of 50 U.S. states specifically legislating medical cannabis and/or CBD use. The DEA is clearly in conflict with the health and well-being of the vast majority U.S. citizens that favor access to medical cannabis and/or CBD. The U.S. state representatives, aware of this fact, blocked the U.S. federal government from enforcing its draconian cannabis laws on any state that has implemented its own cannabis laws. The 2017 federal budget expressly prohibited the Department of Justice from spending any money to prevent states from "implementing their own laws that authorize the use, distribution, possession, or cultivation of medical marijuana." In this context, the cannabinoids THC and CBD as well as genus cannabis plant, when used for medicinal purposes, are protected from federal intervention. CBD derived from industrial hemp, which is non-euphoric, non-intoxicating, and non-psychotropic, has only medicinal purposes and no "recreational" utility.
From a more legal perspective, the DEA is an enforcement agency and not a legislative agency, that is, the DEA is tasked with enforcing the law — not creating or changing existing law. And the law pertaining to cannabinoids extracted from Industrial Hemp vs. marijuana is clear. Although the DEA has defined "marijuana extract" as a schedule I drug and has defined an extract as that containing one or more cannabinoids derived from any plant of the genus Cannabis (including hemp), a judicial court previously ruled otherwise. In Hemp Industries Association vs. DEA, the The United States Court of Appeals for the Ninth Circuit opined: "Appellants manufacture, distribute, or sell comestible items containing oil or sterilized seeds from “hemp†— a species of plant within the genus Cannabis. They challenge two Drug Enforcement Administration (“DEAâ€) regulations that, taken together, would ban the sale or possession of such items even if they contain only non-psychoactive trace amounts of tetrahydrocannabinols (“THCâ€). The DEA asserts that natural, as well as synthetic, THC is included in Schedule I of the Controlled Substances Act (“CSAâ€). We have previously held that the definition of “THC†in Schedule I refers only to synthetic THC, and that any THC occurring naturally within Cannabis is banned only if it falls within the Schedule I definition of “marijuana.†We reiterate that ruling here: in accordance with Schedule I, the DEA’s relevant rules and regulations may be enforced only insofar as they ban the presence of marijuana or synthetic THC." The February 6, 2004 ruling cited above concluded that the DEA "cannot regulate naturally-occurring THC not contained within or derived from marijuana - i.e., non-psychoactive hemp products - because non-psychoactive hemp is not included in Schedule I. The DEA has no authority to regulate drugs that are not scheduled, and it has not followed procedures required to schedule a substance." On September 28, 2004, the Supreme Court declined to hear the DEA's appeal of the case, allowing the Ninth Circuit decision to stand.
On May 22, 2018 the DEA issued an Internal Directive Regarding the Presence of Cannabinoids in Products and Materials Made from the Cannabis Plant. In 2004, the U.S. Court of Appeals for the Ninth Circuit (see above paragraph) "enjoined DEA from enforcing certain regulations with respect to tetrahydrocannabinols (THC).See Hemp Industries Ass'n v. DEA, 357 F.3d 1012 (9th Cir. 2004). The government did not seek Supreme Court review of that decision. In response to various inquiries, DEA hereby issues to DEA personnel the following internal directive on how to carry out their duties in light of the Ninth Circuit's decision." "The Controlled Substances Import and Export Act incorporates the schedules of the CSA. See generally 21 U.S.C. §§ 951-971. Accordingly, any product that the U.S. Customs and Border Protection determines to be made from the cannabis plant but which falls outside the CSA definition of marijuana" (hemp) "may be imported into the United States without restriction under the Controlled Substances Import and Export Act. The same considerations apply to exports of such products from the United States, provided further that it is lawful to import such products under the laws of the country of destination."
At the Federal level, the 2014 Farm Bill allowed individual states to opt in to Hemp Research and Pilot Programs. Thirty-five U.S. states passed Industrial Hemp legislation. In June, 2018, the Senate version of the 2018 Farm Bill passed specifically with a hemp amendment that would officially remove hemp from the DEA’s list of controlled substances. The House version of the bill similarly passed on December 11, 2018, and was signed by the President on December 20, 2018. Federal law is finally up to date with most U.S. State laws regarding the cultivation and use of Industrial Hemp (Cannabis Sativa plant species of less than 0.3% THC). All Hemp-derivatives, extracts and cannabinoids (including CBD) have been officially removed from the federal list of controlled substances, and Hemp has become a legal crop to cultivate in the United States. Previously, legal Hemp was primarily imported in sterilized form (incapable of germination) from European farms. Marijuana-derived cannabinoids (including CBD) remain federally controlled substances (DEA Schedule 1 Drug).
A single oral dose of 5 mg THC in a lipophilic base is rated as placebo dose with respect to acute perceptible psychotropic and physical effects: i.e. it cannot be distinguished from a placebo. Available scientific evidence suggests that a single dose of 5 mg THC and a daily accrued dose of 10 mg THC do not cause acute perceptible effects or chronic detrimental effects on health. Using a safety factor of 10, this leads to a pharmacologically innocuous daily THC intake of 1 mg for a healthy adult. Generally, the THC content of hemp food is so low that psychotropic effects can be excluded with certainty.
Are there other natural foods that come from regulated substances? Yes. Poppy seeds contain narcotics. Poppy seeds are produced by the opium poppy plant and are used to add flavor to many baked goods like bagels and muffins. Poppy seeds contain trace but detectable amounts of the drugs morphine and codeine which are psychoactive compounds. The federal Controlled Substances Act of 1970 lists the poppy species papaver somniferum as a Schedule II drug but specifically exempts papaver somniferum seeds. The trace THC content in hemp is as innocuous as the trace opiod content in (unregulated) poppy seeds.
Be aware that due consideration must be given to the source of any cannabinoid product concerning safety and accuracy. A Research Letter in the November 7, 2017 edition of the Journal of the American Medical Association titled "Labeling Accuracy of Cannabidiol Extracts Sold Online" found that only 30.95% of CBD products purchased online (84 products from 31 companies) were accurate labeled as to their CBD content. This is one reason a highly reputable company with bonafide lab tested products for purity, potency, and accuracy, such as HempMeds Real Scientific Hemp Oil (RSHO), is recommended.
b. How CBD (cannabidiol) Works 
The Short Answer:
CBD interacts with natural chemical messengers and receptors throughout the body to help regulate and balance many important physiological functions.
The Long Answer:
Cannabinoids such as CBD and THC have profound physiological effects on people because the human body has its own an internal system of Cannabinoid Receptors. Two such receptors are called CB1 and CB2. The CB1 receptors are predominant in the brain and nerve system and the CB2 receptors are predominant in organs and cells that function as part of the immune and lymphoid system, but both types of receptors can be found throughout the body and are members of the superfamily of G-protein-coupled receptors.
The reason people have Cannabinoid Receptors in the first place is because the human body makes its own cannabinoids. These are called endocannabinoids (endogenous=from within). Two main naturally occurring endocannabinoids are Anandamine and 2-AG and they interact with cannabinoid receptors CB1 and CB2 (other endocannabinoids include N-arachidonoyl-ethanolamine, N-arachydonoyldopamine, noladin ether and virodhamine). Anandamine binds on the CB1 receptors and 2-AG can be active at both CB1 and CB2 receptors. The actions of the endocannabinoids on the CB1 and CB2 receptors are inhibited by certain enzymes (FAAH, MAGL) which breakdown Anandamine and 2-AG to stop or moderate their action, which is how the endocannabinoid system modulates its activity.
The human body endocannabinoids, cannabinoid receptors, and metabolizing enzymes are called the endocannabinoid system. The current understanding of the endocannabinoid system is that it strives to maintain homeostasis (balance) in the body through the use of chemical messengers to regulate major body functions including sleep, mood, pain, cognition, appetite, metabolism, temperature, memory, immune activity, digestion, inflammation, microcirculation, and reproduction, and therefore has a profound effect on health and well-being. According to studies, the possibility of modulating the activity of the endocannabinoid system holds therapeutic promise in a wide range of conditions, including mood and anxiety disorders, Parkinson’s and Huntington’s disease, neuropathic pain, multiple sclerosis and spinal cord injury, as well as cancer, atherosclerosis, myocardial infarction, stroke, hypertension, glaucoma, obesity/metabolic syndrome, and osteoporosis.
In addition to endocannabinoids made in the body, there are phytocannabinoids made in cannabis plants, of which THC and CBD are the major phytocannabinoids. In the human body, THC (the phytocannabinoid) mimics Anandamine (the endocannabinoid) in binding to CB1 receptors, therefore affecting the brain and nerve system (thinking, behavior, motor control, sedation, appetite). THC is not quickly broken down by the same enzymes that modulate the action of Anandamine. This all explains the psychoactive nature of THC and the prolonged "high" experienced by users of THC.
In contract, CBD does not bind to CB1 or CB2 receptors, but instead 1) inhibits the enzyme that breaks down Anandamine and 2) indirectly promotes 2-AG synthesis – thus increasing endocannabinoid signaling. So the phytocannabinoid CBD is not psychoactive in binding to CB1 like THC and is akin to 2-AG as an agonist of CB2 receptor expression in the organs and cells of the immune and lymphoid system. Because a wide variety of health problems associated with the function of the endocannabinoid system are thought to be related to endocannabinoid deficiency, the supplementation of CBD could help correct this deficiency and improve a number of conditions associated with endocannabinoid system dysfunction through CBD's unique properties as an endocannabinoid modulator. There are numerous clinical studies that suggest exactly this:
CBD up-regulates the activation of the STAT3 transcription factor - anti-inflammatory function
CBD induces apoptosis in activated immune cells, suppress of cytokines and chemokines at inflammatory sites and upregulate regulatory T cells - anti-inflammatory function
CBD demonstrates significant oxidation potential as measured by cyclic voltametry (an electrochemical method for investigating antioxidant compounds, assessing antioxidant capacity, and measuring electrochemical index)
CBD prevents hydroperoxide-induced oxidative damage as well as or better than other antioxidants (including vitamins C and E)
CBD exerts an immunoregulatory effect in activated memory T cells by suppressing proinflammatory Th17-related transcription, promoting T cell exhaustion/tolerance, enhancing IFN-dependent anti-proliferative program, hampering antigen presentation, and inducing antioxidant milieu resolving inflammation - anti-inflammatory function and gene transcription modulator function in autoimmune diseases
CBD modulates activated astrocytes (the most numerous cells populating the central nervous system) which play a significant role in maintaining ion and metabolic balance, debris cleavage, and proper neuronal activity – aids in recovery from neuropathological, neurodegenerative, and neuroinflammatory conditions
CBD represents the first non-toxic exogenous agent that can significantly decrease Id-1 expression in metastatic breast cancer cells leading to the down-regulation of tumor aggressiveness - anti-tumoral function
CBD interacts with Vanilloid receptors - pain modulation function
CBD interacts with Adenosine receptors - anti-anxiety and analgesic function
CBD interacts with Serotonin receptors - mood/stress management function
CBD interacts with GABA receptors - anti-convulsant and anti-anxiety function
CBD prevents glutamate neurotoxicity - antioxidant/neuro-protective function
CBD significantly decreases oxidative stress - oxidation is any chemical reaction that moves electrons, sometimes giving rise to highly reactive oxidants called free radicals (an oxygen containing molecule that has one or more unpaired electrons). Oxidative stress occurs when the body cannot counteract or detoxify the harmful effects of free radicals through neutralization by antioxidants. Oxidation destabilizes and damages cell membranes, cellular proteins, lipids and DNA. An overload of free radicals has been linked to many diseases including deterioration of the eye lens, inflammatory arthritis of the joints, damage to nerve cells in the brain (Parkinson's and Alzheimer's disease), neurodegenerative diseases including ALS, Huntington's disease, Depression, and Multiple sclerosis, acceleration of the ageing process, increased risk of coronary heart disease, and certain cancers triggered by damaged cell DNA.
In summary, according to rigorous scientific research, changes in endocannabinoid levels and/or CB2 receptor expressions have been reported in almost all diseases affecting people ranging from cardiovascular, gastrointestinal, liver, kidney, neurodegenerative, psychiatric, bone, skin, autoimmune, lung disorders to pain and cancer, suggesting that modulating CB2 receptor activity with selective CB2 receptor agonists (such as CBD) holds great therapeutic potential for these pathological conditions.
b. How CBD (cannabidiol) Works - Sources of information:
Cannabinoids in the management of difficult to treat pain https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2503660
Pathways and gene networks mediating the regulatory effects of cannabidiol, a nonpsychoactive cannabinoid, in autoimmune T cells https://jneuroinflammation.biomedcentral.com/articles/10.1186/s12974-016-0603-x
The Profile of Immune Modulation by Cannabidiol (CBD) Involves Deregulation of Nuclear Factor of Activated T Cells (NFAT) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2748879/
Modulation of Astrocyte Activity by Cannabidiol, a Nonpsychoactive Cannabinoid www.mdpi.com/1422-0067/18/8/1669/pdf
Cannabidiol and Δ9-tetrahydrocannabinol are neuroprotective antioxidants https://www.ncbi.nlm.nih.gov/pmc/articles/PMC20965/
Cannabinoids as antioxidants and neuroprotectants https://upload.wikimedia.org/wikipedia/commons/f/f0/US-patent-6630507.pdf
Cannabinoids as novel anti-inflammatory drugs https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2828614/
CBD: How It Works https://www.alchimiaweb.com/blogfr/wp-content/uploads/2015/11/CBDiary21-CBD-How-It-Works.pdf
Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3316151/
The direct actions of cannabidiol and 2-arachidonoyl glycerol at GABAA receptors https://www.ncbi.nlm.nih.gov/pubmed/28249817
The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research https://www.nap.edu/read/24625/chapter/4
Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes https://www.ncbi.nlm.nih.gov/pubmed/21175579
Are cannabidiol and Δ9-tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4301686/#sd2
Molecular Cancer Therapeutics http://mct.aacrjournals.org/content/6/11/2921.short
MedLinePlus-Cannabidiol https://medlineplus.gov/druginfo/natural/1439.html
In a research paper titled "Selective Cannabinoids for Chronic Neuropathic Pain: A Systematic Review and Meta-analysis" the authors concluded that based on the quality of the evidence included in this review and the strength of effect, it can be recommended that selective cannabinoids may be used as adjunct analgesics in patients with neuropathic pain syndromes (Anesthesia & Analgesia: November 2017 - Volume 125 - Issue 5 p 1638-1652).
